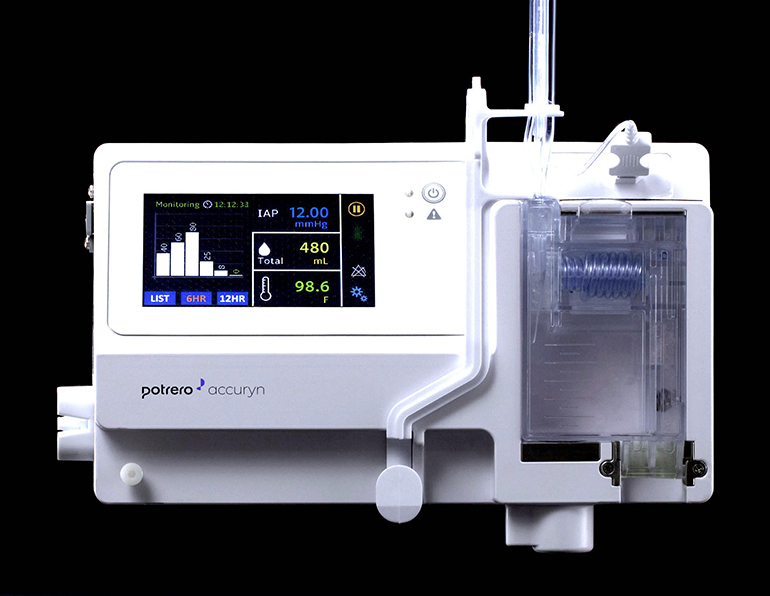
Potrero Medical, Inc. announced today that it received 510 (k) clearance through the Food and Drug Administration (FDA) for the company’s Accuryn™ Monitoring System, which transforms the traditional urinary catheter into a next-generation diagnostic tool for patients requiring tight fluid management, including patients at risk of sepsis and acute kidney injury.
Accuryn™ provides accurate, real-time urine output (UO), intra-abdominal pressure (IAP), and temperature measurement. By automatically clearing the drain line, Accuryn™ enables accurate UO and reduced urinary retention – a known risk for catheter associated urinary tract infection (CAUTI). Accuryn™ is also designed to improve clinical workflow by eliminating manual intervention of drain line blockages and by providing out-of-the box EMR integration.
“Urine output monitoring is a very important field that has stagnated for decades. Based on my personal experience using Accuryn™ in our burn unit, I can say that this device establishes a new state of the art,” Said Bruce Friedman, clinical care co-director of the Joseph M. Still Burn Center. “Accuryn™ is able to eliminate urinary outflow obstruction that is ubiquitous in current urine drainage systems. Solving this problem significantly improves the accuracy and diagnostic value of urine output monitoring and also prevents urinary retention- a major risk factor for CAUTI.”
Potrero Medical, Inc. has demonstrated superiority of Accuryn™ in a multicenter randomized clinical study of Accuryn™ against the current gold standard device. The company is currently engaged in a multi-center study of Accuryn™ to detect sepsis and critical illness (PRESCIENT), which is projected to enroll up to 100 patients in the United States.
Daniel Burnett, MD, Founder and CEO of Potrero Medical stated, “Sepsis and Acute Kidney injury accounted for over $25 Billionin US healthcare costs in 2011 and these costs are growing unsustainably. The Accuryn device will provide actionable data to clinicians which will enable them to improve patient outcomes and to reduce the financial burden of these critical illnesses.”
Potrero Medical will exhibit the Accuryn Monitor at the American Burn Association 48th Annual meeting at Caesar’s Palace, Las Vegas, NV between May 3rd-6th 2016. For more information, please visit http://www.ameriburn.org.
The Accuryn Monitoring System – Important information
The Accuryn Monitoring System is intended for use in the drainage and/or collection of urine, and in the monitoring of urine output and core body temperature, in degrees Fahrenheit and degrees Celsius. The Accuryn Monitoring System is also intended for use in the monitoring of intra-abdominal pressure. The measured pressures can be used as an aid in the diagnosis of intra-abdominal hypertension (IAH) and the associated clinical syndrome of abdominal compartment syndrome (ACS). The Accuryn Sensing Urinary Catheter is a single use device intended for short-term use (less than 30 days).
For information on purchasing Accuryn, please contract Potrero Medical at (415)-926-8616 or sales@potreromed.com.
About Potrero Medical, Inc.:
Potrero Medical, Inc., the latest spinout of medical device incubator Theranova, LLC, is headquartered in San Francisco, CA and was founded with a mission to improve outcomes, reduce costs and expand access to healthcare. For more information about the company, please visit www.potreromed.com.
Potrero Medical, Inc., Contact:
Daniel Burnett
+1 (415) 926-8616